Classification of Elements and Periodicity in Properties Class 11 Notes Chemistry Chapter 3
Why do we need to Classify Elements?
At present 114 elements are known. Efforts to synthesise new elements are continuing. It is very difficult to study such a large number of elements individually and to study their properties separately. The necessity was felt to organise their knowledge in a systematic way by classifying the elements.
Genesis of Periodic Classification: Classification of elements into groups led to the development of Periodic Law and Periodic Table.
J. Dobereiner was the first who made several groups of three elements (Triads). In each case he noticed the middle element of each of the Triads had an atomic weight about halfway between the atomic weights of the other two elements. Also, the properties of the middle element were in between these of the other two elements. He referred to them as the Law of Triads. It worked for only a few elements.
Dobereiner’s Triads:
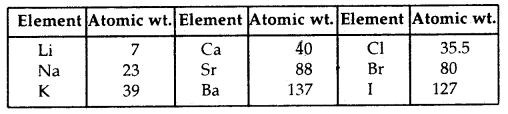
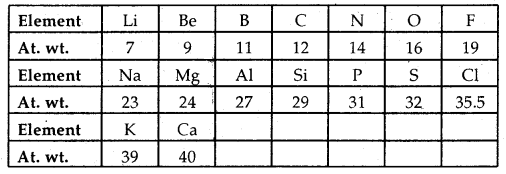
J.A. Newlands propounded the Law of Octaves. He arranged the elements in increasing order of their atomic weights and noted that every eighth element has properties similar to the first element like the octaves of music – sa, re, ga, ma, pa, dha, nee, sa Newland’s Law of Octaves seemed to be true only for elements up to calcium.
Newlands Octaves:
Russian chemist Mendeleev and German chemist Lothar Meyer working independently, let to the Periodic Law as we know it today. Mendeleev succeeded in arranging the elements in vertical columns called groups and horizontal rows called periods in his table based upon the increasing order of their atomic masses. He gave his well- known law:
”The properties of the elements are a periodic function of their atomic weights”. It was called Mendeleev law. He realised that .some of the elements did not fit in with his scheme of classification if the order of atomic weights was strictly followed. He ignored the order of atomic weights and placed the elements -with similar properties together. For example, iodine with a lower atomic weight than that of tellurium (Group VI) was placed in Group VII along with other halogens F, Cl, Br because of similarities in properties.
He left some gaps for elements that were yet undiscovered. For example, both gallium (Ga) and germanium (Ge) were unknown at his time: He left the gap below aluminium and a gap below silicon and called these elements Eka-Aluminium and Eka-Silicon. The boldness of Mendeleev’s quantitative predictions and their eventual success made him and his Periodic Table famous. Mendeleev’s periodic table published in 1905 is shown.
Characteristics of Mendeleev’s Periodic Table:
It consists up of
- Eight vertical columns called groups. Except for the VIII group, each group is. further subdivided in A and B. This subdivision is made on the basis of difference in their properties.
- Six horizontal row called periods.
Significance of Mendeleev’s Periodic Table
- Instead of studying properties of elements separately, they can be studied in groups containing elements with the same properties. It led to the systematic study of the elements.
- Prediction of new elements. At his time only 56 elements were known. He left blank spaces or groups for unknown elements.
- Mendeleev’s periodic table corrected the doubtful atomic weights.
Defects in the Mendeleev’s Periodic Table
- Hydrogen was placed in group IA. However, it resembles both groups IA elements (alkali metals) and group VII A (halogens). Therefore the position of hydrogen in the periodic table is Anomalous or Controversial.
- Anomalous pairs of elements. Some elements with higher atomic weight like Argon (39.9) precede potassium (39.1) with lower atomic weight.
- Based upon atomic weights, isotopes of an element could not be assigned to different groups. They have been assigned only one position in a group.
- Some dissimilar elements are grouped together while some similar elements are placed in different groups. For example, alkali metals in group IA which are highly reactive are in the same group as coinage metals like Cu, Ag, Au of group IB. At the same time, certain chemically similar elements like Cu (group IB) and Hg (group IIB) have been placed in different groups.
- Position of elements of group VIII. No proper place has been allotted to nine elements of group VIII which have been arranged in three triads without any justification.
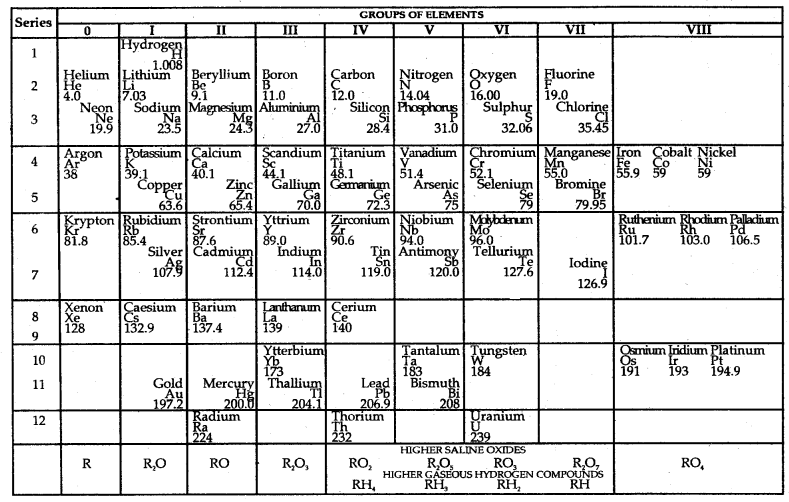
Mendeleev’s Periodic Table published in 1905
Modern Periodic Law and the present form of the Periodic Table.
Modern Period Law: The physical and chemical properties of the elements are periodic functions of their atomic number.
According to the recommendations of IUPAC, the groups are numbered from 1 to 18 replacing the older notation of groups 0, 1A, II A
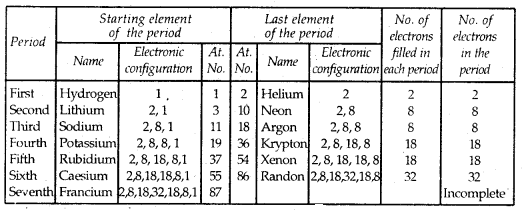
There are 7 periods. The first period contains 2 elements. The subsequent periods contain 8, 8, 18, 18 32 elements respectively. The 7th period is incomplete and like the 6th period would have a theoretical maximum of 32 elements. In this form of Periodic Table, the elements of both the sixth and seventh periods (lanthanoids and actinoids respectively) are placed in Separate panels at the bottom.
Long Form of Periodic Table
General characteristics of the long form of the Periodic table:
1. There are in all, 18 vertical columns or 18 groups in the long-form periodic table.
2. These groups are numbered from 1 to 18 starting from the left.
3. There are seven horizontal rows called periods in the long-form periodic table. Thus, there are seven periods in the long-form periodic table.
| The first period contains | 2 elements | Shortest period |
| The second & Third period contains | 8 elements each | Short period |
| The fourth & Filth period contains | 18 elements each | Long-period |
| Sixth periods contains | 32 elements | Longest period |
| The seventh period contains | It is incomplete | Incomplete period |
4. The elements of group 1, 2 .and 13 to 17 are called the main group elements. These are also called typical or representative or normal elements.
5. The elements of group 3 to 12 are called transition elements.
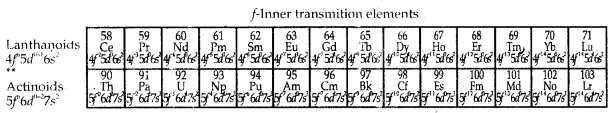
6. Elements with atomic number 58 to 71 (Ce to Lu) occurring after lanthanum (La) are called Lanthanides. Elements with atomic numbers 90 to 103 (Th to Lr) are called Actinides. These elements are called f-block elements and also inner-transition elements.
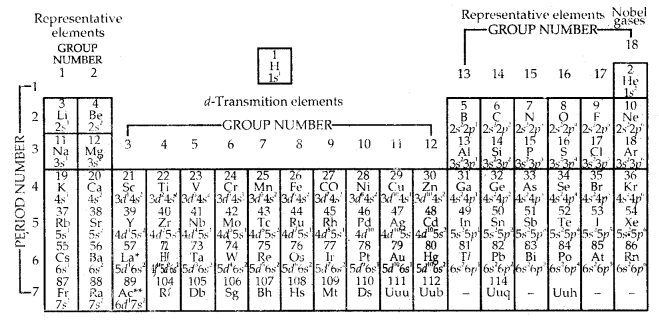
Long-form of the Periodic Table of the EIenient with their atomic numbers and ground state outer
electronic configurations. The groups are numbered i-18 in accordance with the 1984 IUPAC recommendations. this notation replaces the old numbering scheme of IA-VIIA, 4dIII, IB—VIIB and I) for the elements.
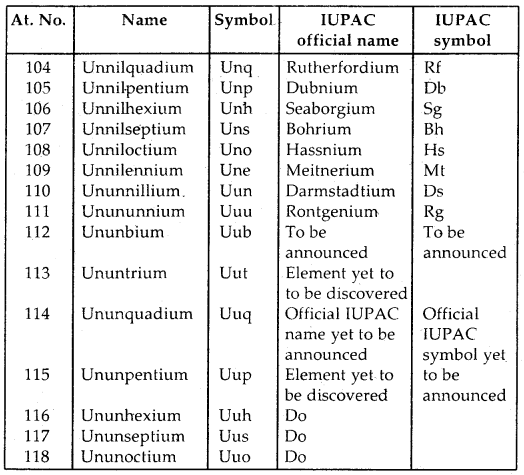
Nomenclature of the Elements with Atomic No. > 100
Table: Nomenclature of elements with atomic number above 103
Notation for IUPAC Nomenclature of Elements
Electronic Configuration of the Elements and Periodic Table: In the long-form periodic table, the elements are arranged in the order of their atomic numbers. An atomic number of an element is equal to the number of protons inside the nucleus of its atom. In an atom, the number of electrons is equal to the number of protons (hence equal to the atomic number). As a result, there is a close connection between the electronic configurations of the elements and the long form of the periodic table.
Table: The relationship between the electronic configuration of electrons and their positions in the periodic
Electronic Configuration in Periods: The period indicates the value of n- for the outermost or valence shell. In other words, a successive period in the Periodic Table is associated with the filling of the next higher principal energy level (n = 1, n = 2, etc.). It can be readily seen that the number of elements in each period is twice the number of atomic orbitals available in the energy level that is being filled.
→ The first period (n = 1) starts with the filling of the lowest level (1s) and therefore has two elements hydrogen (1s1) and helium (1s2) when the first shell (K) is completed. The second period (n = 2) starts with lithium and the third electron and has the electronic configuration 1s22s2. Starting from the next element boron, the 2p orbitals are filled with electrons when the L shell is complete at neon (2s22p6).
→ Thus there are 8 elements in the second period. The third period (n = 3) begins at sodium, and the added electron enters a 3s orbital. Successive filling of 3s and 3p orbitals gives rise to the third period 8 elements from sodium to argon. The fourth period (n = 4) starts at potassium, and the added electrons fill up the 4s orbital.
→ Now you may note that before the 4p orbital is filled, filling up of 3d orbitals becomes energetically favourable and we come from scandium (Z = 21) which has the electronic configuration 3d1, 4s2. The fourth period ends at krypton with the filling up the ‘Ip orbitals. Altogether we have 18 elements in this fourth period. The fifth period (n = 5) beginning with rubidium is similar to the fourth period and contains the 4d transition Serietarting at yttrium (Z = 39). This period ends al xenon with the filling up of the Sp orbitals.
→ The sixth period (n = 6) contains 32 elements and successive electrons enter 6s, 4f, Sd and 6p orbitals, in the order-filling up of the 4f orbitals begins with caesium (Z = 58) and ends at lutetium (Z = 71) Lo give 4f-inner transition series which is called lanthanoid sêries. The seventh period (n = 7) is similar to the sixth period with the successive filling up of the 7s, 5f, 6d and 7p orbitals and includes most of the man-made radioactive elements.
→ This period will end at the elements with atomic number 118 which would belong to the noble gas family. Filling up of the 5f orbital after actinium (Z = 80) gives the 5f-inner transition series known as the actinoid series. The 4f- and 5f- transition series of elements are placed separately in the Periodic Table to maintain its structure and to preserve the principle classification by keeping elements with similar properties in a single column.

Thus it can be seen that the properties oían element have periodic dependence upon its atomic number and not on relative atomic mass. All the elements in the same group have the same number of electrons (ns1 in alkali metals) in their valence shells and thus have, same properties.
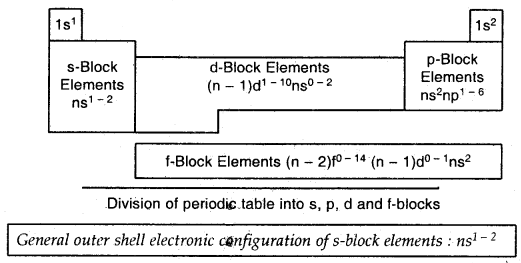
Electronic Configurations and Types of Elements s-, p-, d-, f-Blocks:
The Aufbau (build-up) principle and the electronic configuration of atoms provide a theoretical foundation for periodic classification. The elements in a vertical column of the Periodic Table constitute a group or family and exhibit similar chemical behaviour. This similarity arises because these elements have the same number and same distribution of electrons in their outermost orbitals. We can classify the elements into four blocks viz., s-block, p-block, d- block and f-block depending on the type of atomic orbitals that are being filled with electrons. We notice two exceptions to this categorization.
Strictly, helium belongs to the s-block but its positioning in the p-block along with another group of 18 elements is justified because it has a completely filled valence shell (1s2) and as a result, exhibits properties characteristic of other noble gases. The other exception is hydrogen. It has alone s-electron and hence can be placed in group-1 (alkali metals). It can also gain an electron to achieve a noble gas arrangement and hence it can behave similarly to group 17 (halogen family) elements. Because it is a special case, hydrogen is placed separately at the top of the Periodic Table.
→ The s-Block Elements: The elements of group 1 (alkali metals) and group 2 (alkaline earth metals) which have ns1 and ns2 outermost electronic configuration belong to the s-Block Elements. They are all reactive metals with low ionization enthalpies. They lose the outermost electron(s) readily to form + lions (in the case of alkali metals) or + 2ion (in the case of alkaline earth metals). The metallic character and the reactivity increase as we go down the group. Because of high reactivity, they are never found pure in nature. The compounds of the s-block elements, with the exception of those of lithium and beryllium, are predominantly ionic.
→ The p-Block Elements: The p-Block Elements comprise those belonging to groups 13 to 18 and these together with the s-block elements are called the Representative Elements or Main Group Elements. The outermost electronic configuration varies from nsTnpl to ns2np6 in each period. At the end of each period is noble gas elements with a closed valence shell ns2np6 configuration.
All the orbitals in the valence shell of the noble gases are completely filled by electrons and it is very difficult to alter this stable arrangement by the addition or removal of electrons. The noble gases thus exhibit very low chemical reactivity Preceding the noble gas family are two chemically important groups of non-metals. They are the halogens (Group 17) and the chalcogens (Group 16).
These two groups of elements have high negative electron gain enthalpies and readily add one or two electrons respectively to attain the stable noble gas configuration. The non-metallic character increases as we move from left to right across a period and the metallic character increases as we go down the group.
→ The d-Block Elements (Transition Elements): These are the elements of Group 3 to 12 in the centre of the Periodic Table These are characterised by the filling of inner (n – 1) d orbitals by electrons and are therefore referred to as d-Block Elements. These elements have the outer electronic configuration (n – 1)d1-10ns1-2. They are all metals. They mostly form coloured ions, exhibit variable valence (oxidation .states), paramagnetism and are often used as catalysts.
However, Zn > Cd and Hg which have, the electronic configuration, (n – 1)d10ns2 do not show most of the properties of transition elements. In a way, transition metals form a bridge between the chemically active metals of s-block elements and the less active metals of Groups 13 and 14 and thus take their familiar name “transition elements”.
→ The f-Block Elements (Inner-Transition Elements): The two rows of elements at the bottom of the Periodic Table, called the Lanthanoids, Ce (Z = 58) – Lu (Z = 71) and Actinoids, Th (Z = 90) – Lr (Z = 103) are characterised by the outer, electronic configuration (n – 2)f1-14(n – 1)d0-1 ns2. The last electron added to each element is an f-electron.
These two series of elements are hence called the inner- transition elements (f-Block Elements). They are all metals. Within each series, the properties of the elements are quite similar. The chemistry of the early -actinoids .is more complicated than the corresponding lanthanoids, due to a large number of oxidation states possible for these actinoid elements.
Actinoid elements are radioactive. Many of the actinoid elements have been made only in nanogram quantities or even less by nuclear reactions and their chemistry is not fully studied. The elements after uranium are called transuranium elements.
The Lanthnoids show predominantly an oxidation state of + 3 (with a few exceptions + 2 or + 4) whereas Actinoids show variable oxidation states.
Metals, Non-Metals and Metalloids: The elements can be divided into Metals and Non-metals. Metals comprise more than 78% of all known elements and appear on the left side of the Periodic Table. Metals are usually solids at room temperature (mercury is an exception; gallium and caesium also have very low melting point 300K and 302K respectively). Metals usually have high melting and boiling points. They are good conductors of heat and electricity. They are malleable (can be flattened into thin sheets by hammering) and ductile (can be drawn into wires). In contrast, non-metals are located at the top right-hand side of the Periodic Table.
In fact, in a horizontal row, the property of elements change from metallic on the left to non-metals on the right. Non-metals are usually solids or gases at room temperature with low melting and boiling points (boron and carbon are exceptions). They are poor conductors of heat and electricity. Most non-metallic solids are brittle and are neither malleable nor ductile. The elements become more metallic as we go down a group; the non-metallic character increases as one goes from left to right across the Periodic Table.
The change from metallic to non-metallic character is not abrupt. The elements (e.g.r silicon, germanium, arsenic, antimony and tellurium) bordering this line and running diagonally across the Periodic Table show properties that are characteristic of both metallic and non-metals. These elements are called Semi-Metals or Metalloids.
Periodic Trends in Properties of Elements:
1. Ionization Enthalpy: The amount of energy required to remove the most loosely bound electron from an isolated gaseous atom in its ground state is called ionisation enthalpy.
X (g) → X(g) + e–
The energy required to remove the first electron is known as the first ionization enthalpy and the second electron is the second ionization enthalpy and so on. The second ionization enthalpy is always greater than The first ionization enthalpy because once an electron is removed, it becomes a positive ion and its nucleus has increased attraction for electrons. This makes it more difficult to remove the second electron.
Ionization enthalpy decreases from top to bottom in a group and increases horn left to right in a period. Thus Cs has the lowest ionization enthalpy and fluorine has the highest ionization enthalpy. Thus IE3 > IE2 > IE1. IE of Li > Na > K > Rb > Cs and IE of Li in 2nd Period is lowest, whereas that of Na is highest.
The ionization enthalpy depends on two factors:
- The attraction of electrons to the nucleus.
- The repulsion of electrons from each other.
Screening or Shielding effect: A valence electron in a multi-electron atom is pulled by the nucleus And repelled by the other electrons in the core (inner shells). Thus the effective pull on the electron will be the pull due to the positive nucleus, less the repulsion due to core electrons.
The effective repulsive effect to the core electrons is called the screening or shielding effect. For example, the 2s electron in lithium is shielded by the inner core of Is electrons. As a result valence electron experiences a net positive charge less than + 3. In general, shielding is effective when the orbitals in the inner shells are completely filled.
2. Electron Gain (Enthalpy ΔegH): It is defined as the enthalpy change when a neutral gaseous atom takes up extra electrons to form an anion.
X(g) + e– → X– (g)
The value of electron gain enthalpy depends upon the atomic size, nuclear charge etc. with an increase in size, the electron gain enthalpy decreases as the nuclear attraction decreases. Thus electron gain enthalpy generally decreases in going from top to bottom in a group (F < Cl > Br > I). In a period from left to right, the electron gain enthalpy generally increases due to an increase in nuclear charge.
Thus halogens have very high electron affinity. However, the electron affinity of fluorine is less than that of chlorine. This is because fluorine has a small atomic size (only two shells). Also, electron repulsion is more in the case of fluorine, because of mutual electron-electron repulsions.
Factors affecting electron gain enthalpy:
- Atomic size: Smaller the atom, the greater is the magnitude of the electron gain enthalpy. This is because the added electron can go closer to the nucleus and as a result releases more energy.
- Effective nuclear charge: Greater the nuclear charge, the larger is the magnitude of electron gain enthalpy. This is because the electron would experience stronger attraction by virtue of a higher nuclear charge.
- Electronic configuration of the atom: An atom with a stable electronic configuration has a little or no tendency to add another electron. As a result, such elements have zero, or nearly zero electron gain enthalpy.
Periodic Variation of Electron gain enthalpy:
1. Variation of electron gain enthalpy in a group: The magnitude of the electron gain enthalpy of elements decreases in going from top to bottom in a group. However, the electron gain enthalpy of fluorine is lower than that of chlorine.
On moving from top to the bottom of a group,
(a) the-atomic size increases and
(b) the nuclear charge also increases.
The effect of these factors is opposite to each other. In group, the effect of an increase in the atomic size outweighs the effect of the increased nuclear charge. As a result, the tendency to accept an electron in its valency. shell and hence the magnitude of electron gain enthalpy decreases as we go down the group.
2. Variation of electron gain Enthalpy in a period: The magnitude of electron gain enthalpy increases across a period is going from left to right.
On moving from left to right in a period, the size of the atoms decreases, and the effective nuclear charge increases. Both these factors increase the force of attraction exerted by the nucleus on the electrons. As a result, the atom has a greater tendency to gain an extra electron from outside and therefore, the magnitude of electron gain enthalpy increases in going from left to right. However, some elements in each period show an exception to such periodicity. For example, Be, Mg, N, P and noble gases show very low, or even zero electron gain enthalpies.
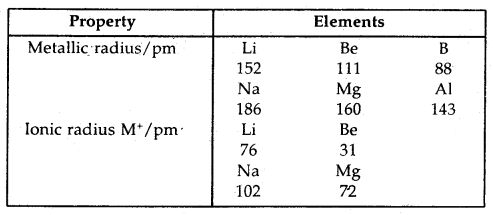
Periodic Variation of Atomic and Ionic Radii: Atomic radius is one half of the distance between the nuclei of two identical atoms in a molecule bonded by a single bond. In a period, as, we move from left to right, the number of shells remain the same but as more electrons are added the nuclear charge increases.
This results in an increase in the number attraction for the electrons, which in turn brings about a decrease in the radius of the elements in a period. Thus, the radius decreases along a period (from left to right). However, in a group from top to bottom; the number of shells increases, therefore, the radius also increases, e.g., rLi < rNa < rK < rRb < … etc.
→ Ionic Radius: Ionic radius may be defined as “the effective distance from the centre and*nucleus of an ion up to which it has an influence on its electron cloud”.
→ Variation of Ionic radii in a group: The ionic radii of the ions belonging to the same group of elements and having identical charges, increases in going from top to bottom in a group. For example, the radii of the monovalent alkali metal ions increase from Li+ to Cs+.
→ Variation of the radii of isoelectronic ions: Atoms/ions of different elements having the same number of electrons are called isoelectronic ions. In other words, the ions which have the same number of electrons but different nuclear charges are called isoelectronic ions. The size of such ions depends upon the nuclear charge (as the total number of electrons remains the same).
Thus N3- > O2- > F– > Na+ > Mg2+ > Al3+
Thus for isoelectronic ions, the ionic size decreases as the nuclear charge increases.
Cations are always smaller than their parent atoms because of
- the disappearance of the valence shell in some cases
- increase in effective nuclear charge.
Thus Na+ < Na and Mg2+ < Mg.
Anions are always bigger in size than their parent atom. With the addition of one or more electrons to an atom, the effective nuclear charge per electron decreases. The electron cloud thus increases leading to an increase in the size of the anion.
Thus, F– > F; Cl– > Cl; O2- > O; S2- > S and so on.
→ Electronegativity: The ability of an atom in a chemical compound to attract the shared pair of electrons towards itself is called Electronegativity. It is not a measurable quantity, unlike IE and electron gain enthalpy. L Pauling gave numerical values of electronegativities to elements. F has been given a value of 4.0 arbitrarily, the highest in the periodic table. Electronegativity values are not constant. Electronegativity generally decreases from top to bottom in a group and increases across a period from left to right.
Electronegativity values (Pauling scale) across a period

Within a Group
| Group-1 | Group-17 |
| Li = 1.0 | F = 4.0 |
| Na = 0.9 | Cl = 3.5 |
| K = 0.8 | Br = 2.8 |
| Rb = 0.8 Cs = 0.7 |
I = 2.5 At = 2.2 |
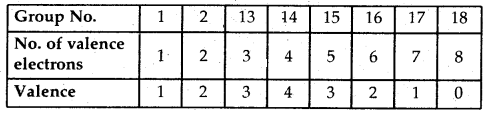
Periodicity of Valence: Valence is the most characteristic property of the elements and is based upon electronic configurations. The valence of representative elements is usually (though not necessarily) equal to the number of valence electrons arid or equal to eight minus the number of outermost electrons.
→ Anomalous Behaviour of Second Period Elements:
The first element of each of the groups 1 (lithium) and 2 (beryllium) and groups 13-17 (boron to fluorine) differs in many respects from the other, members of their respective group. For example, lithium unlike other alkali metals, .and beryllium unlike other alkaline earth metals, form compounds with pronounced covalent character, the other members of these groups predominantly form ionic compounds.
In fact, the behaviour of lithium and beryllium is more similar to the second element of the following group i.e., magnesium and aluminium, respectively. This sort of similarity is commonly referred to as a diagonal relationship in the periodic properties.
→ What are the reasons for the different chemical behaviour Of the first members of a group of elements in the s- and p- blocks compared to that of the subsequent members in the same group?
The anomalous behaviour is attributed to their small size) large charge/radius ratio and high electronegativity of the elements. In addition, the first member of the group has only four valence orbitals (2s and 2p) available for bonding, whereas the second member of the groups has nine valence orbitals (3s, 3p, 3d). As a consequence of this, the maximum covalency of the first member of each group is 4, whereas the other members of the groups can expand their valence shell to accommodate more than four pairs of electrons.
Furthermore, the first member of p-block elements displays greater ability to form pπ – pπ multiple bonds to itself (e.g., C = C, C ≡ G, N = N, N ≡ N) and to other second period elements (e.g., C = O, C = N, C ≡ N, N = O) compared to subsequent members of the same group.
1. Lothar-Meyer Arrangement of Elements: When plotting a graph between the atomic volumes (gram atomic weight divided by density) and atomic weights of the elements he observed that the elements with similar properties occupied similar positions on the curve.
2. Mendeleev’s Periodic Law: The physical and chemical properties of elements are a periodic function of their atomic weights.
3. Moseley’s/Modem Periodic Law: The physical and chemical properties of the elements are a periodic function of their atomic numbers.
4. Periodicity of Properties of Elements: According to Modem Periodic Law the properties of the elements are repeated after certain regular intervals when these elements are arranged in order of their increasing atomic numbers. These regular intervals 2, 8, 8, 18, 18, 32 are called Magic Numbers.
5. Cause of Periodicity: The cause of periodicity in properties is the repetition of similar electronic configurations of the valence shells after certain regular intervals.
6. Groups: The vertical columns of elements in the periodic table are called Groups.
7. Periods: The horizontal rows of elements in the periodic table are called Periods:
→ There are 18 groups and 7 periods in the Modern Periodic Table
1. s-block Elements: These are the elements in which the last electron enters the s-subshell of the valence orbit.
2. p-Block Elements: The p-block elements are those in which the last electron enters the p-subshell of the vaLence orbit. The elements of groups 13, 14, 15, 16, 17, 18 (excluding helium) in which p-orbitals are being progressively filled in are called p-block elements. Since each group has five elements, therefore, ¡ri all, there are 30 p-block elements in the periodic table.
The elements of the 18th group are called noble gases.
General outer shell electronic configuration of p-block elements: ns2 np1 6
3. d-Block Elements: Elements in which the last electron enters any one of the five d-orbitals of their respective penultimate, i.e., (n – 1)the shells are called d-bock elements.
Transition Elements: Since the properties of these elements are midway between those of s-block and p-block elements, they are called Transition Elements.
General outer shell electronic confìguration of d-block elements: (n—1)d1-10 n2.
Zn, Cd, Hg do not show most of the properties of transition elements.
4. f-Block Elements: Elements in which the last electron enters any onè o the seven f-orbitals of their respective ante-penultimate shells are called f-block elements. In all these elements, the s-orbital of the last shell (n.) is completely filled, the d-orbital of the penultimate (n – 1) shell invariably contains zero or one electron but the J-orbitals of the antepenultimate (n – 2) shell (being lower in energy than d – orbitals of the penultimate shell) gets progressively filled in. Hence
General outer shell electronic configuration.off-block elements: (n – 2)0-14 (n – 1) d0-1 ns2
→ They are called Lanthanides/Lanthanones/Lanthanoids. In their case, the antepenultimate 4/subshell is being filled up.
→ They are called Actinides/Actinones/Actinoids if their antepenultimate 5/subshell is being b lied up.
All the actinoids are radioactive elements.
→ Elements from neptunium to lawrencium (93Np – 103Lr) which have been prepared artificially through nuclear reactions are called Transuranic or Transuranium elements as (hey follow uranium in the periodic table.
→ Metalloids: The elements like silicon, germanium, arsenic, antimony and tellurium (Si, Ge, As, Sb, Te) which show the properties of both metals and non-metals are called Metalloids.
→ Ionization Enthalpy: The minimum amount of energy required to remove the most loosely bound electron from an isolated gaseous atom of an element is called it’s Ionization Energy/Ionization Potential/Ionization Enthalpy.
M (g) + energy → M+ (g) + e– (g)
→ Units of I.E.: electron volts (eV) per atom
or
kilo calories per mole (k cal mol-1)
or
kilo Joules per mole (kj mol-1)
1 eV per atom = 23.06 kcal mol-1
= 96.49 kj mol-1.
→ Successive I.Es.
IE3 > IE2 > IE1
where M (g) + IE1 → M+ (g) + e– (g)
M+ (g) + IE2 → M2+ (g) + e– (g)
M2+ (g) + IE3 → M3+ (g) + e– (g)
→ Variation of IE across a period: It generally increases from left to right in a period.
→ Variation of IE within a group: It generally decreases from top to bottom within a group.
→ Electron Gain Enthalpy of an element may be defined as the energy released when a neutral isolated gaseous atom accepts an extra electron from outside.
X(g) + e– → X– (g); ΔH = ΔegH
→ Variation across a period: In general, it becomes more and more negative from left to right in a period.
→ Variation within a group: In general, it becomes less negative as we move down a group.
→ Atomic Radius: The average distance from the centre of the nucleus to the outermost shell containing electrons.
They are of three types:
- Covalent radius
- Vander Waal’s radius
- Metallic radius
→ Variation across a period: In general it decreases in a period with an increase in atomic number from left to right.
→ Variation within a group: It increases with the increase in atomic number from top to bottom within a group.
→ Ionic Radius: It is defined as the effective distance from the centre of the nucleus of the ion up to which it exerts Its influence on the electronic cloud.
→ Variation within a group: The ionic radius increases as we move down a group.
The size of a cation is always less than its corresponding atom
Na+ < Na
Mg2+ < Mg
The size of an anion is always larger than its corresponding atom
Cl– > Cl
O2- > o
→ Isoelectronic Ions or species: Ions of different elements which have the same number of electrons but the different magnitude of the nuclear charge are called Isoelectronic ions.
Na+, F–, Mg2+ etc. are iso electronic ions.
The ionic radii of isoelectronic ions decrease with the increase in nuclear charge.
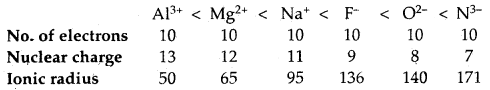
→ Trends in groups and periods: Electronegativity. values for the representative elements increase along period and decrease down the group.
Summary of the Trends in the Periodic Properties of Elements in the Periodic Table
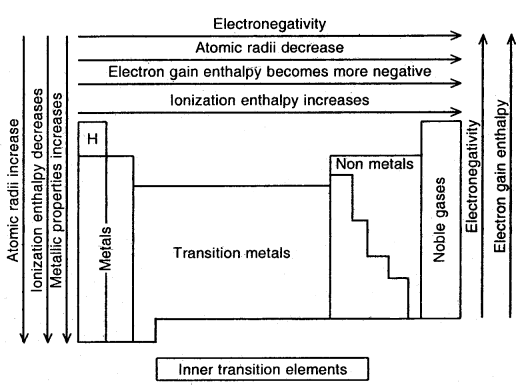
→ Valency: The electrons present in the outermost shell of an atom are called valence electrons and the number of these electrons determine the valence or the valency of the atom. It is because of this reason that the outermost shell is also called the valence shell of the atom and the orbitals present in the valence shell are called valence orbitals.
→ Variation along a period: As the no. of electrons increases from 1 to 8 in representative elements across a period, the valency first increases from 1 to 4 and then decreases to zero in the case of noble gases.
→ Variation within a group: As the no. of electrons remain the same within a group, therefore, all the elements in a group exhibit the same valency.
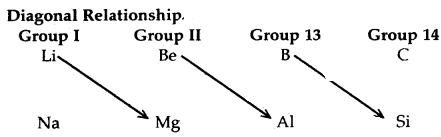
The resemblance of properties of Li with Mg (which is diagonally situated); Be with Al and B with Si is called Diagonal relationship.
It is due to similar polarising power.